CDC issues guidance on prioritizing new RSV treatment for infants
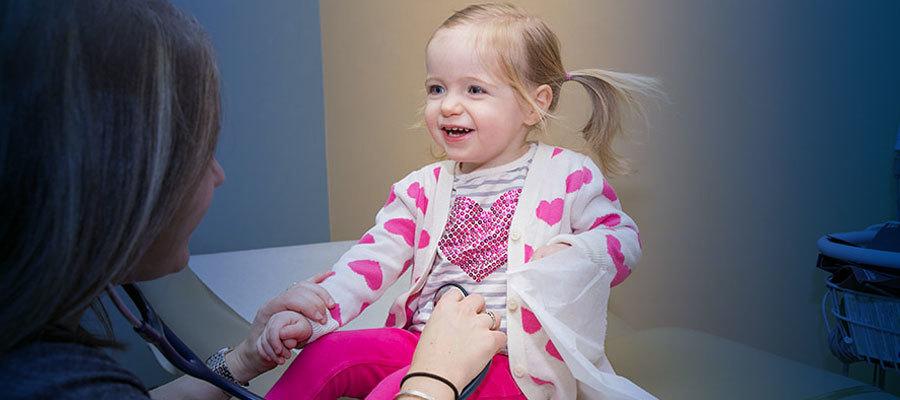
The Centers for Disease Control and Prevention Oct. 23 released interim guidance for clinicians with limited access to the monoclonal antibody nirsevimab, recently approved to prevent respiratory syncytial virus in children aged 2 and under. The guidance calls for prioritizing 100 milligram doses of the treatment for infants under 6 months old and infants at high risk for severe disease due to underlying health conditions, among other recommendations.
Related News Articles
Headline
A recording of a free webinar on maternity care coding hosted by the American Medical Association on June 2 is now available. Registration is required to view…
Headline
Rep. Randy Feenstra, R-Iowa, introduced the Rural Maternity Options for Medical Support Act on May 19. The bill would guarantee that beds used solely for labor…
Headline
Sutter Health is taking a proactive, systemwide approach to maternal care — supporting a range of birth experiences while reinforcing the…
Blog
High-quality maternal care is essential to protecting the health of both mom and baby during birth.Sutter Health is taking a proactive, systemwide approach to…
Headline
Katie Au, M.D., and Katherine Jorda, M.D., directors of the Perinatal Trauma Clinic at Oregon Health & Science University, explore how…
Headline
The U.S. birth rate declined by 1% in 2025, according to preliminary data released by the Centers for Disease Control and Prevention. The cesarean delivery…

